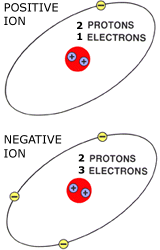
In a crystal of salt, every sodium ion is attracted to all surrounding chlorine atoms, and the chlorine atom is attracted to all surrounding ions. This arrangement of ions is held together by strong electrostatic forces between the oppositely charged ions. When many positive (+ve) and negative (-ve) ions come together, they can arrange themselves in a regular, repeating pattern known as a crystal lattice. Many positive (+ve) and negative (-ve) ions arrange themselves in a structure called a crystal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
